Address: 3-1-24 Matsuyama, Kiyose-shi, Tokyo 204-8533 Japan
Phone: +81-42-493-5711
Facsimile: +81-42-492-4600
URL: http://www.jata.or.jp
『BCG 2nd Edition -TB vaccine- Application against tuberculosis and other diseases』

This book is the only book which summarizes the research on BCG vaccine from the basics to epidemiological studies on effectiveness of the vaccine. Furthermore, the book also covers the topics related to the development of the new tuberculosis vaccine and the application of BCG vaccine to other diseases. We hope that the book will be beneficial to readers including students and expert researchers in the field of tuberculosis.
https://www.jatahq.org/product/bcg-2nd-edition-tb-vaccine-application-against-tuberculosis-and-other-diseases
Finding missing TB cases: Choosing algorithms and tools
[JICA-Net Library] Tuberculosis Control and Community Engagement
A world where no one suffers from tuberculosis.
Our mission is to eliminate TB suffering through development and implementation of comprehensive TB control strategies.
Activities
In order to fulfill our mission, we will carry out the following activities both in and outside Japan.
- Conduct bacteriological and molecular epidemiological studies, including development of new diagnostics, which contribute to the understanding of the biology of Mycobacterium tuberculosis
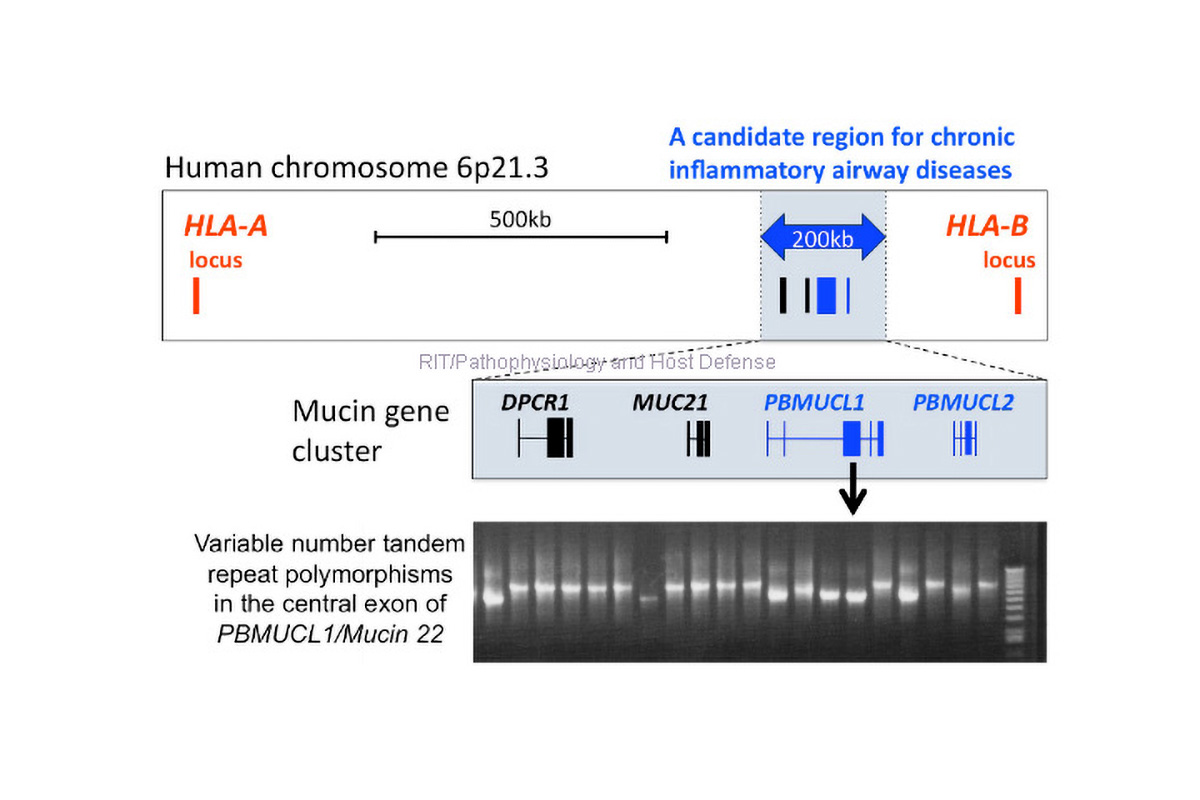
- Conduct basic and translational researches on pathophysiology, host-pathogen interaction and pharmacology against mycobacteria using clinical samples and animal models.
- Conduct epidemiological, operational and clinical researches, which contribute to evidence-based policies for TB prevention, care and control.
- As a supra-national reference laboratory in the Western Pacific Region, provide strategic and technical assistance to national tuberculosis reference laboratories of countries in the region.
- Provide professional consultancy and support to local and national TB control programs, including epidemiological assessment, monitoring and evaluation, and outbreak investigation.
- Contribute to human resource development through organizing and implementing various training courses.